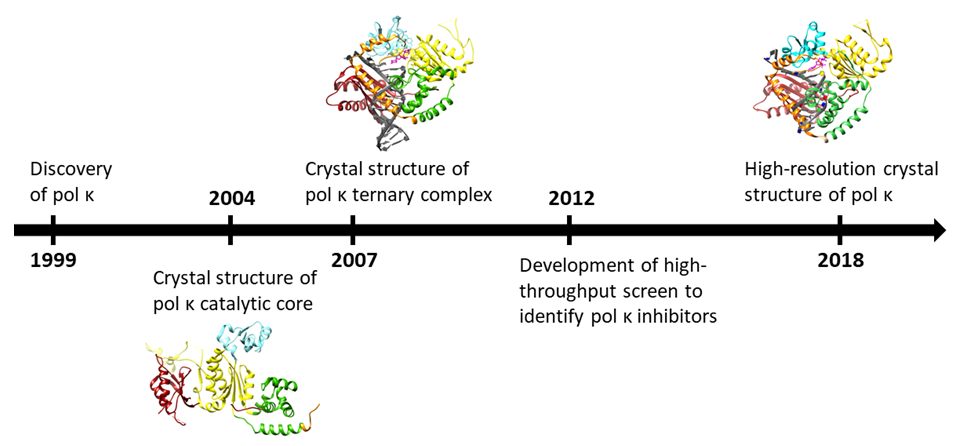
DNA polymerase eta: A potential pharmacological target for cancer therapy - Saha - 2021 - Journal of Cellular Physiology - Wiley Online Library

Model depicting the role of Pol eta in the replication of CFS. (A) Pol... | Download Scientific Diagram
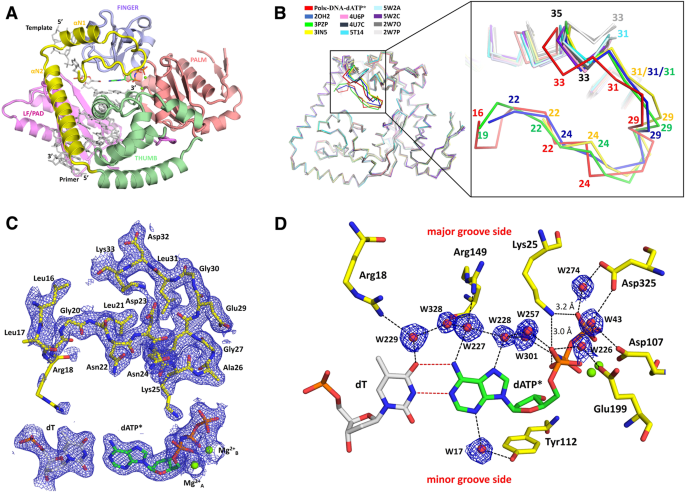
![Structural basis of accurate replication beyond a bulky major benzo[a]pyrene adduct by human DNA polymerase kappa - ScienceDirect Structural basis of accurate replication beyond a bulky major benzo[a]pyrene adduct by human DNA polymerase kappa - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S1568786416303597-fx1.jpg)
Structural basis of accurate replication beyond a bulky major benzo[a]pyrene adduct by human DNA polymerase kappa - ScienceDirect
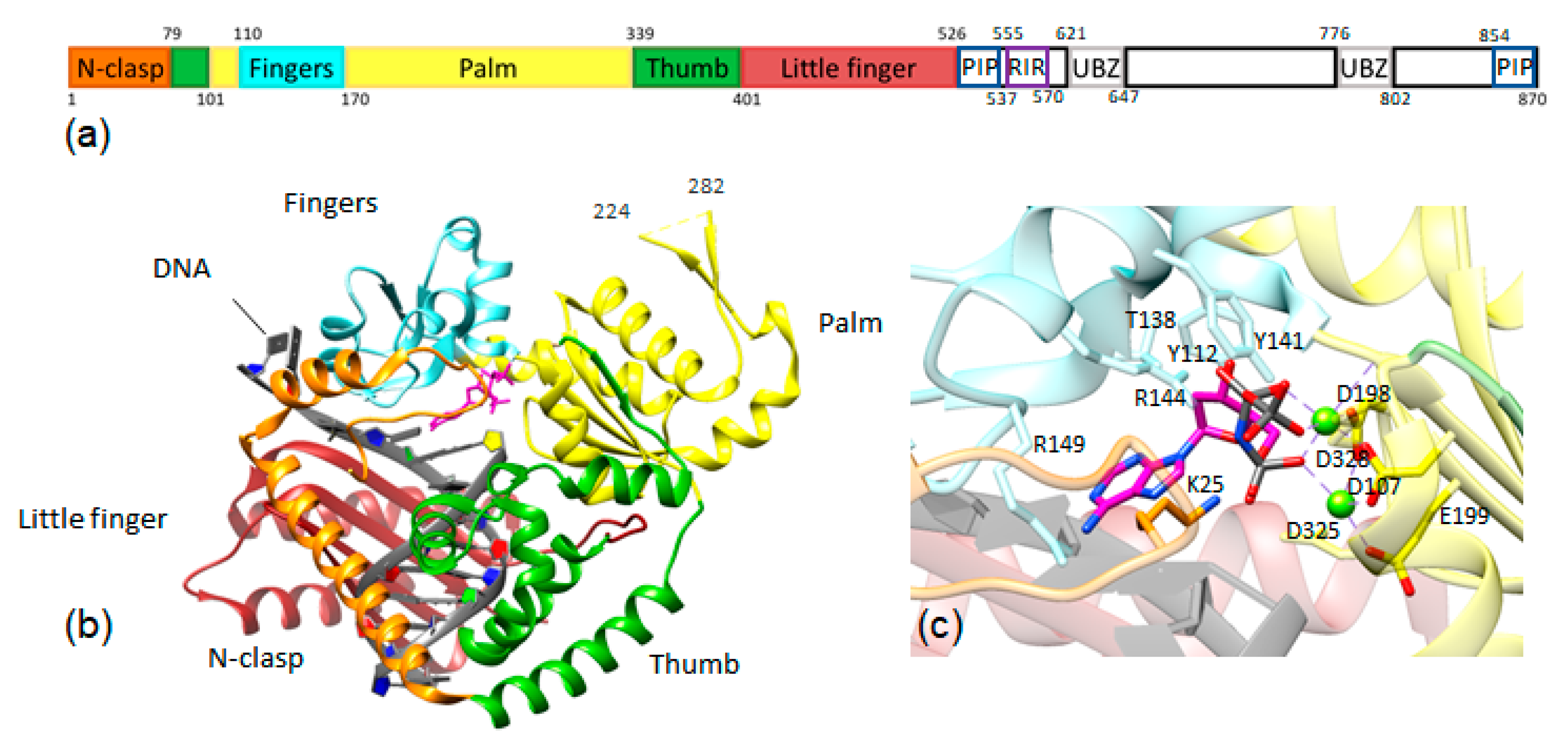
2.0 Å resolution crystal structure of human polκ reveals a new catalytic function of N-clasp in DNA replication | Scientific Reports

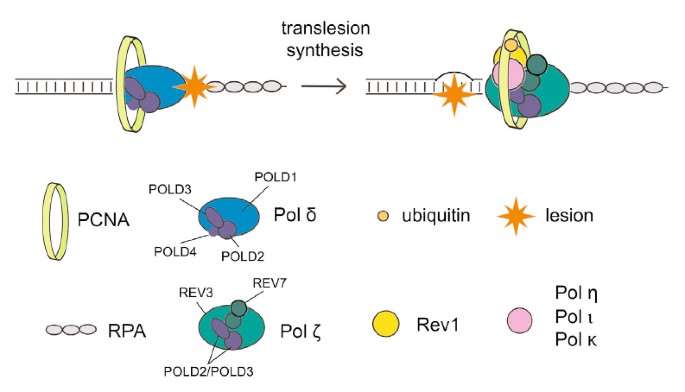
Translesion polymerase eta both facilitates DNA replication and promotes increased human genetic variation at common fragile sites | PNAS

In vivo evidence that DNA polymerase kappa is responsible for error-free bypass across DNA cross-links induced by mitomycin C - ScienceDirect

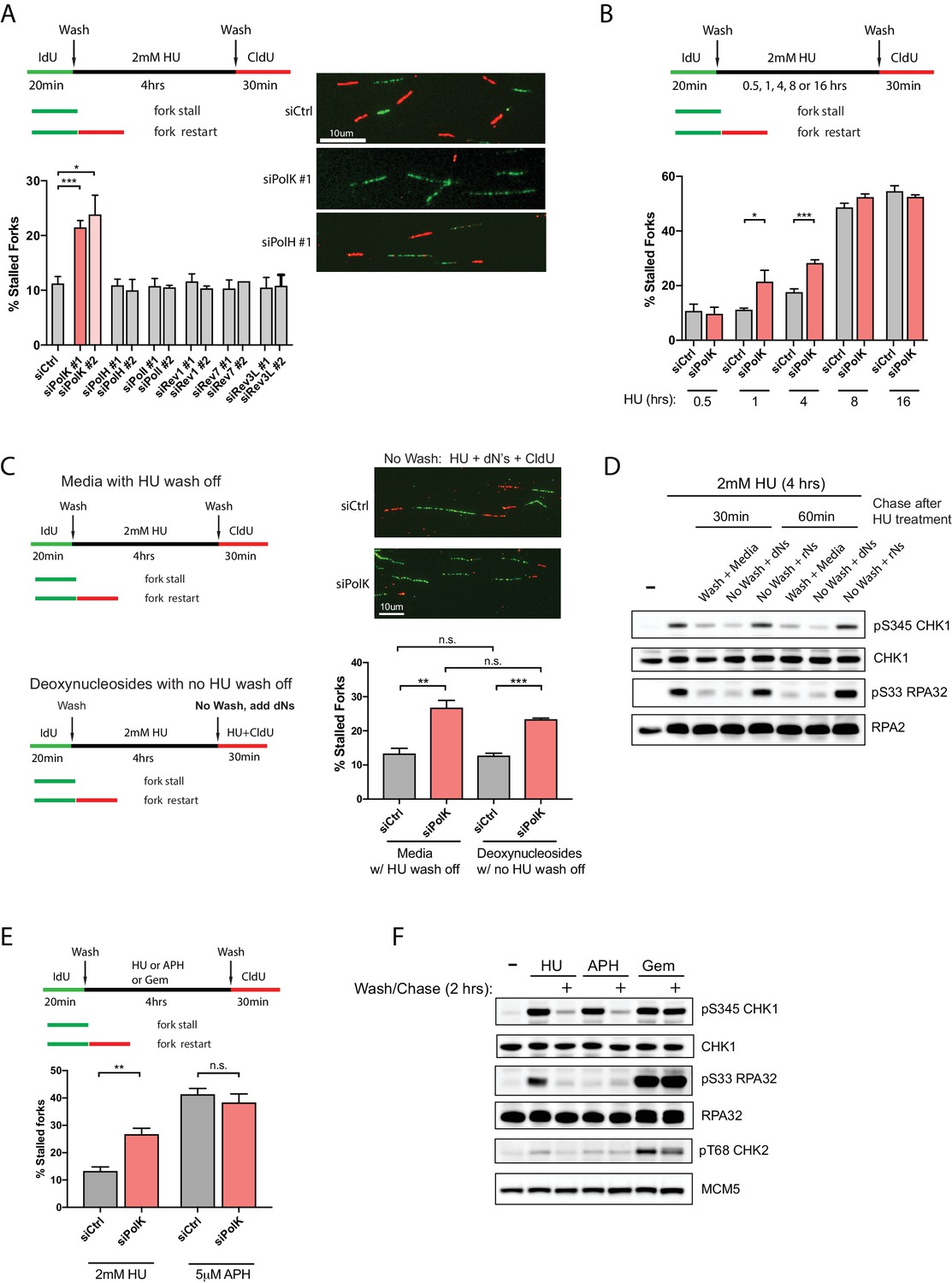
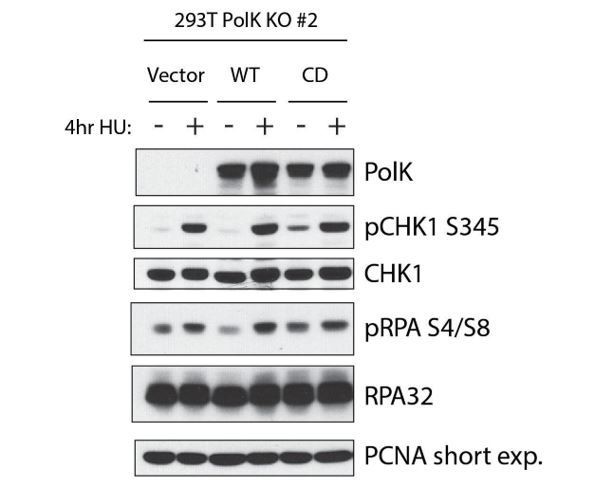
A catalytic-independent function of human DNA polymerase Kappa controls the pool of the Checkpoint Kinase 1 | bioRxiv
DNA Polymerase Eta Participates in the Mutagenic Bypass of Adducts Induced by Benzo[a]pyrene Diol Epoxide in Mammalian Cells | PLOS ONE

Inhibition of Human DNA Polymerases Eta and Kappa by Indole-Derived Molecules Occurs through Distinct Mechanisms | ACS Chemical Biology